Working Principle
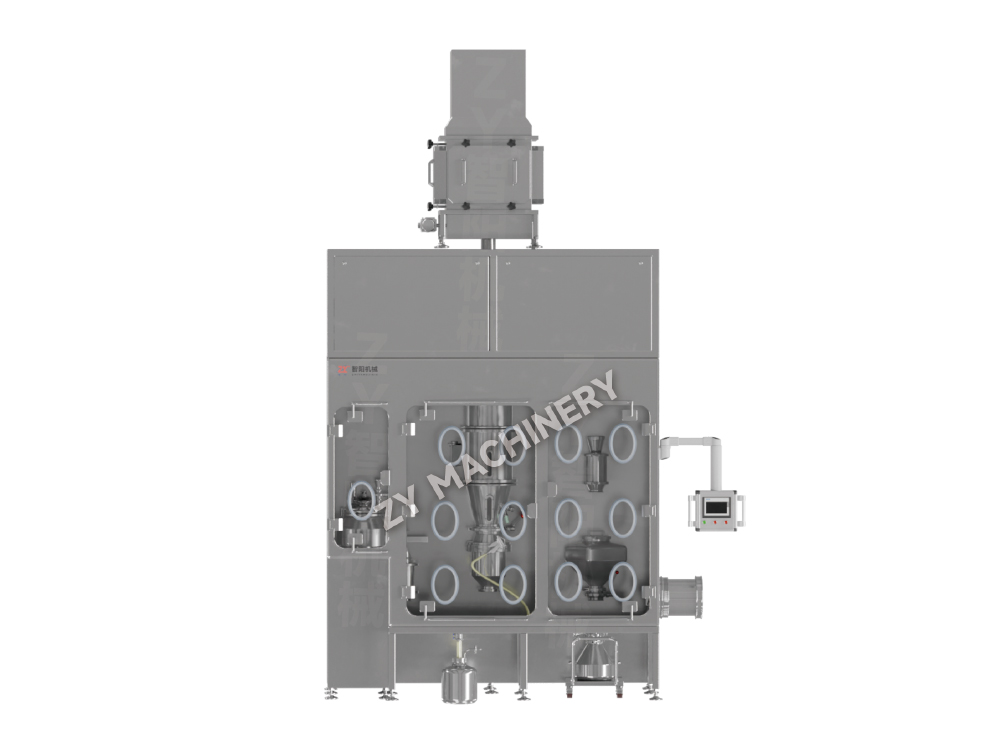
Material is transferred into a sealed isolator via a Rapid Transfer Port (RTP) valve. Within the isolator, the granules or tablets are processed in the coating chamber. Once coating is complete, the finished products are safely transferred out through either a transfer chamber or sleeve system, maintaining containment throughout the process.
BGB HR&D High-Containment, High-Efficiency Coating Machine
The BGB HR&D High-Containment Coating Machine is engineered for applying coatings to highly potent tablets in the pharmaceutical industry. It is suitable for products such as hormones, anticancer medications, and other high-toxicity compounds. The system ensures a safe, efficient, and hygienic coating process that fully complies with GMP standards while maintaining strict operator protection.
Specializes in the design, manufacturing, and process integration of powder processing and oral solid dosage equipment. BGB HR&D High-Containment, High-Efficiency Coating Machine Suppliers and BGB HR&D High-Containment, High-Efficiency Coating Machine Company in China. By combining reliable process know-how, continuous innovation, and proven performance value, we provide standalone machines, modular systems, and complete production lines for customers in pharmaceutical, bio-pharmaceutical, biotechnology, nutraceutical, veterinary, additives, and related industries—from lab and pilot scale to full production.
Founded in 2010, ZY Machinery is process-driven. BGB HR&D High-Containment, High-Efficiency Coating Machine for sale. We design solutions around material characteristics, capacity targets, and site conditions. We do not simply supply machines—we deliver workable process routes and engineering implementation plans to help customers achieve a better balance of consistency, efficiency, reliability, and cost.
-
Sieving Machines as Core Refining Equipment Sieving machines are a fundamental category of refining equipment, designed to transform heterogeneous bulk materials into uniform, high-purity products. By passing material through precisely controlled mesh apertures, these machines remove oversized cont...
READ MORE -
Pharmaceutical vacuum drying equipment achieves rapid, low-temperature drying that preserves API stability, reduces residual solvents to below regulatory limits (typically ≤0.5% or 500 ppm), and enables up to 95% solvent recovery. Data from industrial scale-ups show that switching from conventional ...
READ MORE -
The true capability of high-speed mixing equipment is defined not by motor horsepower alone, but by the tip speed and shear rate generated at the mixing head. A high-speed disperser operating at a tip speed of 4,000–6,000 ft/min can reduce particle agglomerates in a pigment dispersion from 50 µm to...
READ MORE

 English
English Español
Español русский
русский 中文简体
中文简体

 PDF Download
PDF Download